
The development of Immune Checkpoint Inhibitors (ICIs) represents a major advance in the treatment of cancer. ICIs “release the brakes” on T cells that kill cancer cells. Although ICIs have shown great promise, still only a limited number of patients achieve durable response to therapy. A major cause for this is a lack of tumor T-cell infiltration characterized as “cold tumors.” Hence, there is a need for interventions that can convert “cold” non-T-cell infiltrated tumors to “hot” T-cell infiltrated tumors.
When cancer cells are exposed to Lytix's molecules, the cancer cell membrane will disintegrate, resulting in a necrotic cell death and destruction of intracellular organelles such as mitochondria. As a result, “danger signals” (danger associated molecular patterns, DAMPs) and a wide spectrum of tumor antigens will be released from cancer cells, facilitating an optimal activation of dendritic cells and subsequent anti-cancer immune response. This process leads to an increased influx of T cells into the tumor. By reprogramming of the tumor microenvironment through the eradication of local immunosuppressive cells followed by infiltration of cytotoxic T cells, one may reinstate systemic anticancer immune responses, hence eliminating all deposits of remaining cancer cells.
Lytix's molecules address heterogeneity of the tumor as they exerts its activity through membranolytic effect and are equally active against both therapy-resistant and therapy-sensitive cancer cell.
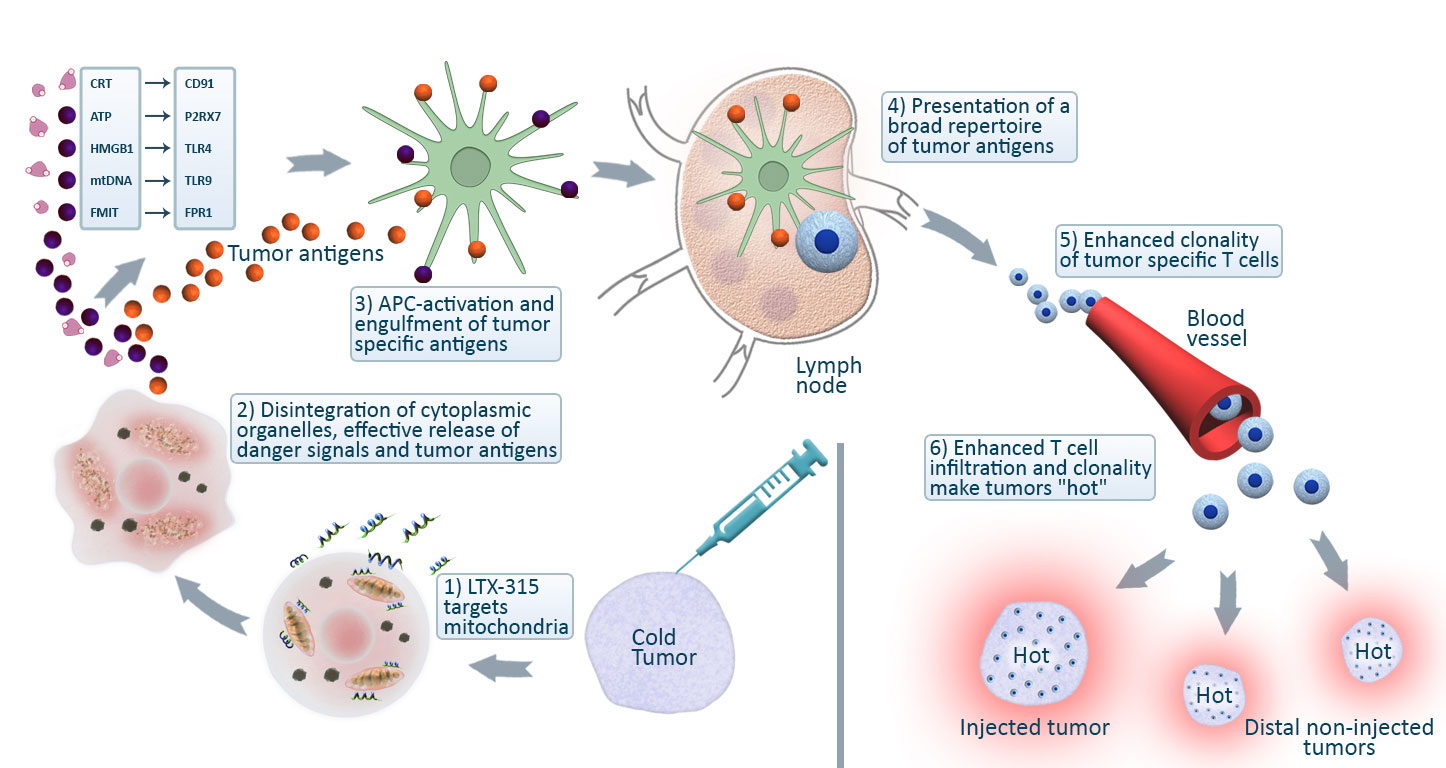
Figure 1: Dual mechanism of action for intratumoral treatment with LTX-315
LTX-315
Lytix Biopharma has developed a first-in-class 9-mer peptide, LTX-315, modified from the natural host-defense peptide lactoferricin. LTX-315 exerts its activity through membranolytic effect, and selectivity for cancer cells results from the cationic amino acid side chains of LTX-315 interacting with the abundant anionic membrane components present at higher levels on cancer cells than in normal cells.
Intra-tumoral (local) treatment with LTX-315 results in growth inhibition, complete regression and a long-lasting tumor specific immune response in a wide variety of experimental models. An abscopal effect on non-treated lesions has been observed in both experimental metastasis models and in clinical trials. The treatment efficacy is associated with increased infiltration of T cells into the tumor post-treatment and reprogramming of the tumor microenvironment, including decreases in the local immunosuppressive regulatory T-cell and myeloid-derived suppressor cell populations.
Consistent with the LTX-315-mediated modulation of the tumor microenvironment, the diversity and clonality of T-cell clones in both circulation and within the tumor expands post-treatment as demonstrated by immuno-sequencing of the T-cell receptor b-chain repertoire.
The ability of LTX-315 to increase T-cell infiltration and T-cell clonality indicates that LTX-315 has the potential to convert “cold” tumors to “hot” tumors, which positions LTX-315 as an ideal combination partner for other cancer therapies, including ICIs.
LTX-401
LTX-401 is a novel small molecule designed for local treatment of deep-seated tumors and it shares similar pharmacological properties with the peptide LTX-315. LTX-401 has been shown to exert strong anticancer effects in a wide variety of experimental models, including liver cancer, in which it stimulates T-cell infiltration and subsequent regression of tumors.
When combined with ICIs in an experimental model, LTX-401 demonstrated a strong abscopal effect on distant lesions, an effect not seen with ICIs alone in these models.
LTX-401 has shown to be safe and well tolerated in preclinical safety studies.
LTX-315 is an oncolytic immunotherapy. Time lapse study with confocal microscopy shows that the human melanoma cell (A547) is killed within minutes by LTX-315. (Courtesy of L.M. Eike and B. Sveinbjørnsson, UiT)
Presentation of LTX-315 – the discovery and its mode of action.
